#1 ALM SOFTWARE FOR PHARMA DEVELOPMENT
Enable Agile & Digital Engineering.
Enforce Traceability & Compliance.
Accelerate Your Timelines.
Simplify your complex pharmaceutics and biomedical product development, and begin ensuring end-to-end traceability and compliance across industry standards with Visure.
- Most cost-effective
- Access All Features
- 30-Day Trial


1,000+ Highly Regulated Organizations Trust Visure














Guarantee Pharma Product Development Standards and Regulations Compliance with Industry Templates
Transform pharma product development work across all engineering teams with a modern platform that is designed for safety-critical standards and regulations in accordance with the Gamp 5 guide. This results in speed time to market and low validation costs without sacrificing compliance, or product quality.
- GAMP 5
- FDA 21 CFR Part 11
- FMEA
- DFMEA

Achieve Project Success with an All-in-One AI-powered Pharma ALM Software
 Get faster requirements elicitations by authoring, reading, and editing items in a single view without losing visual context in the overall document.
Get faster requirements elicitations by authoring, reading, and editing items in a single view without losing visual context in the overall document.
Achieve a balance between atomic requirements management and traceability, all while maintaining a familiar document-style approach.
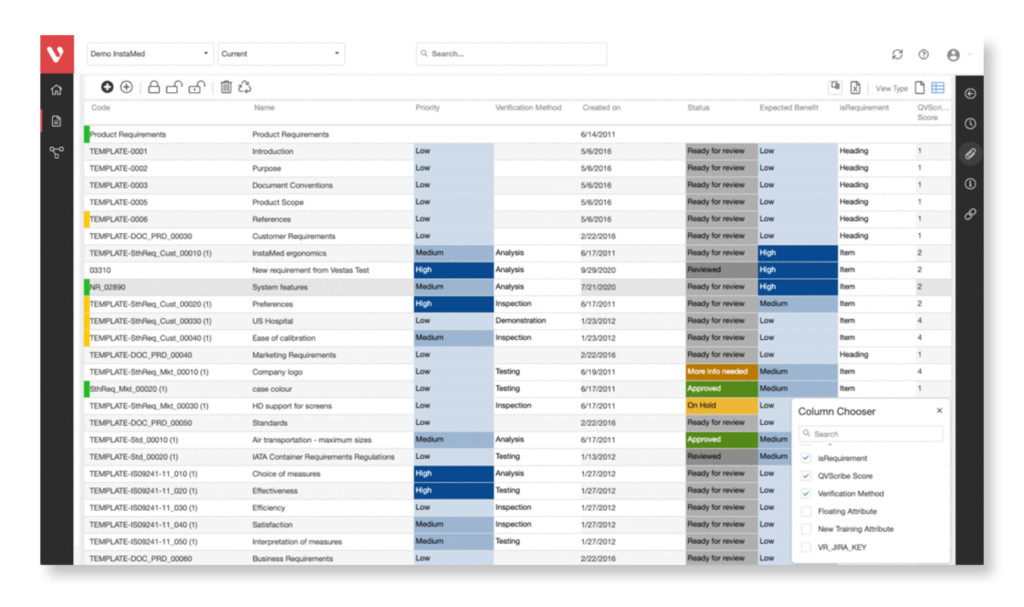
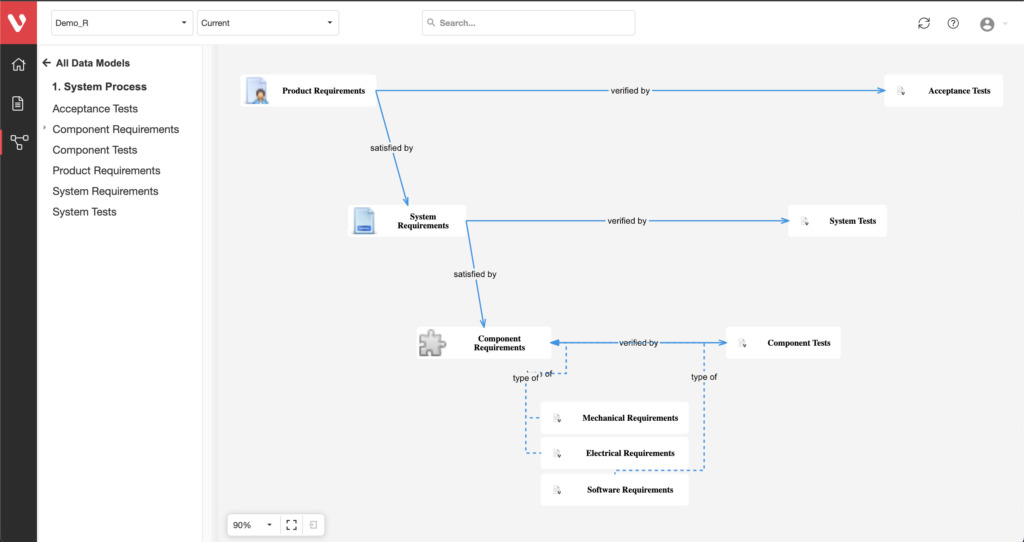
 Gain end-to-end traceability for Risk, Requirements, and Tests. Implement a robust change management process to uncover process interdependencies and maintain project success.
Gain end-to-end traceability for Risk, Requirements, and Tests. Implement a robust change management process to uncover process interdependencies and maintain project success.
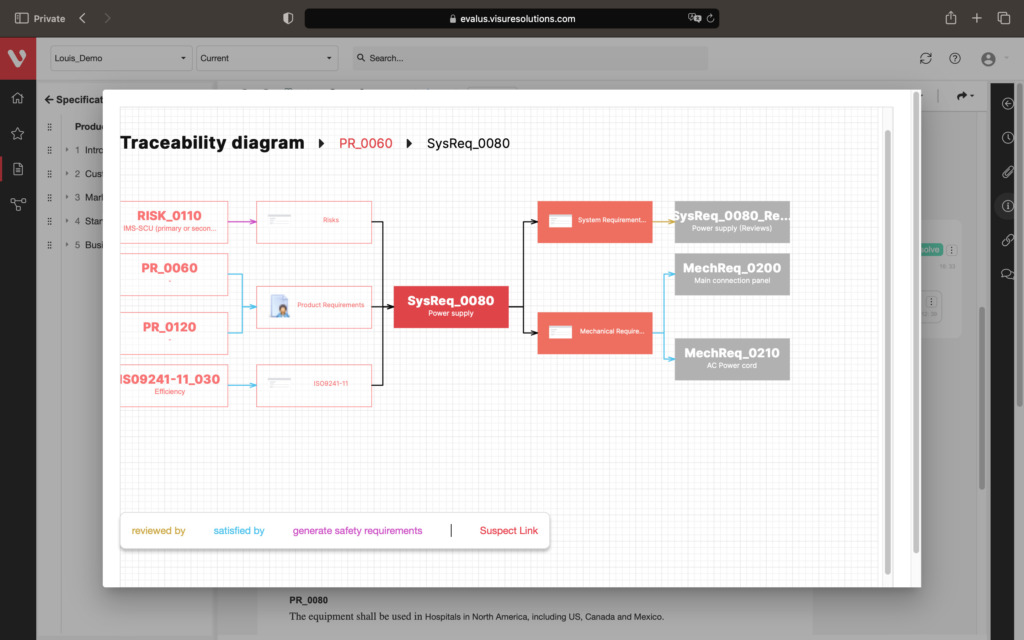
Effortlessly explore upstream and downstream relationships, anticipate change impact, monitor cross-project relationships, detect potential link issues during changes, and visualize relationship rules across projects to understand their organizational impact and reach.
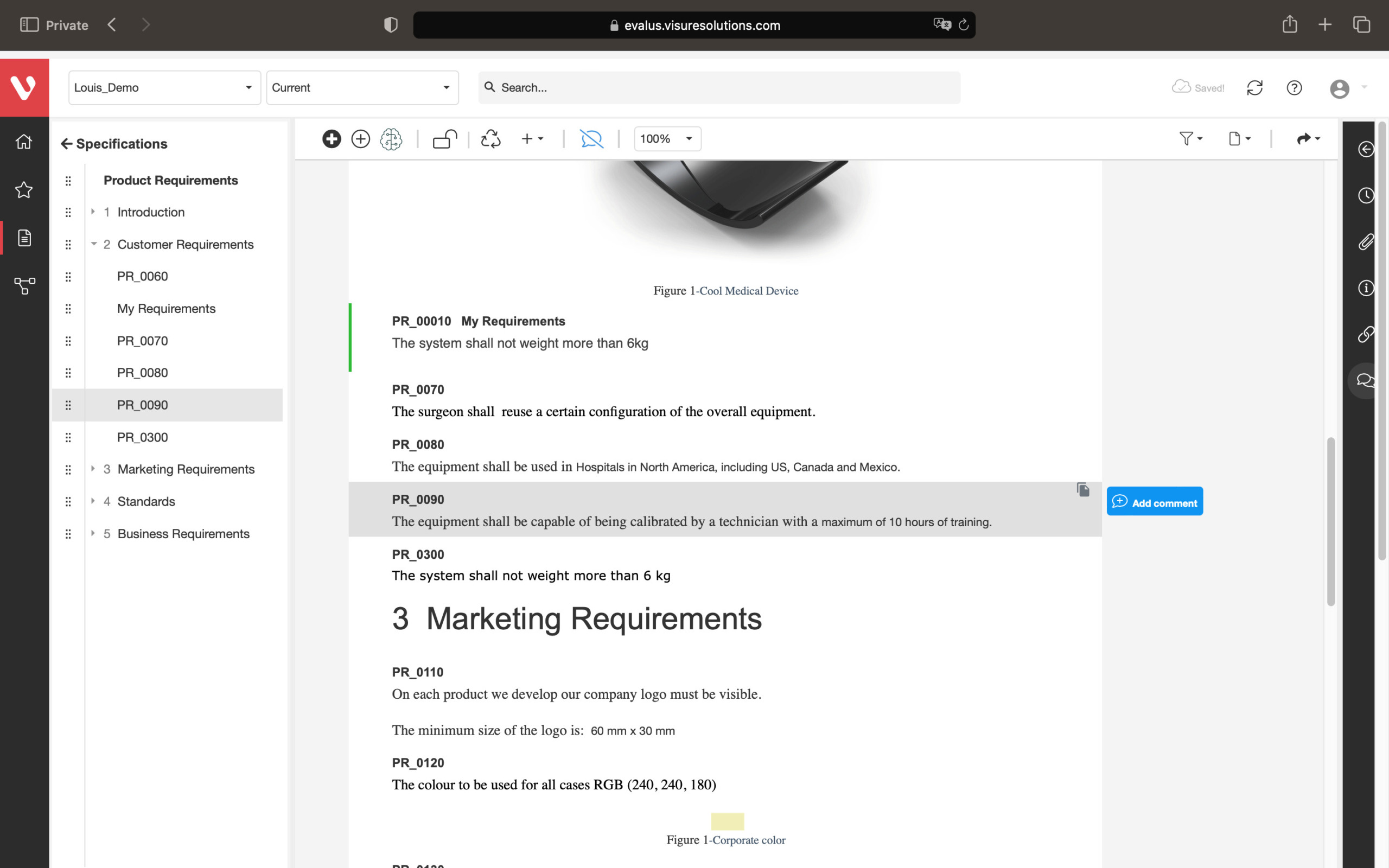 Reduce risk and simplify compliance audits. Visure facilitates early and ongoing risk analysis, allowing you to identify, estimate, and assess risks.
Reduce risk and simplify compliance audits. Visure facilitates early and ongoing risk analysis, allowing you to identify, estimate, and assess risks.
Keep your risk analysis current with real-time data, improve coverage by tracing open risks to requirements, build a comprehensive risk management profile using PHA and FMEA techniques, and guarantee quality and safety in intricate product development.
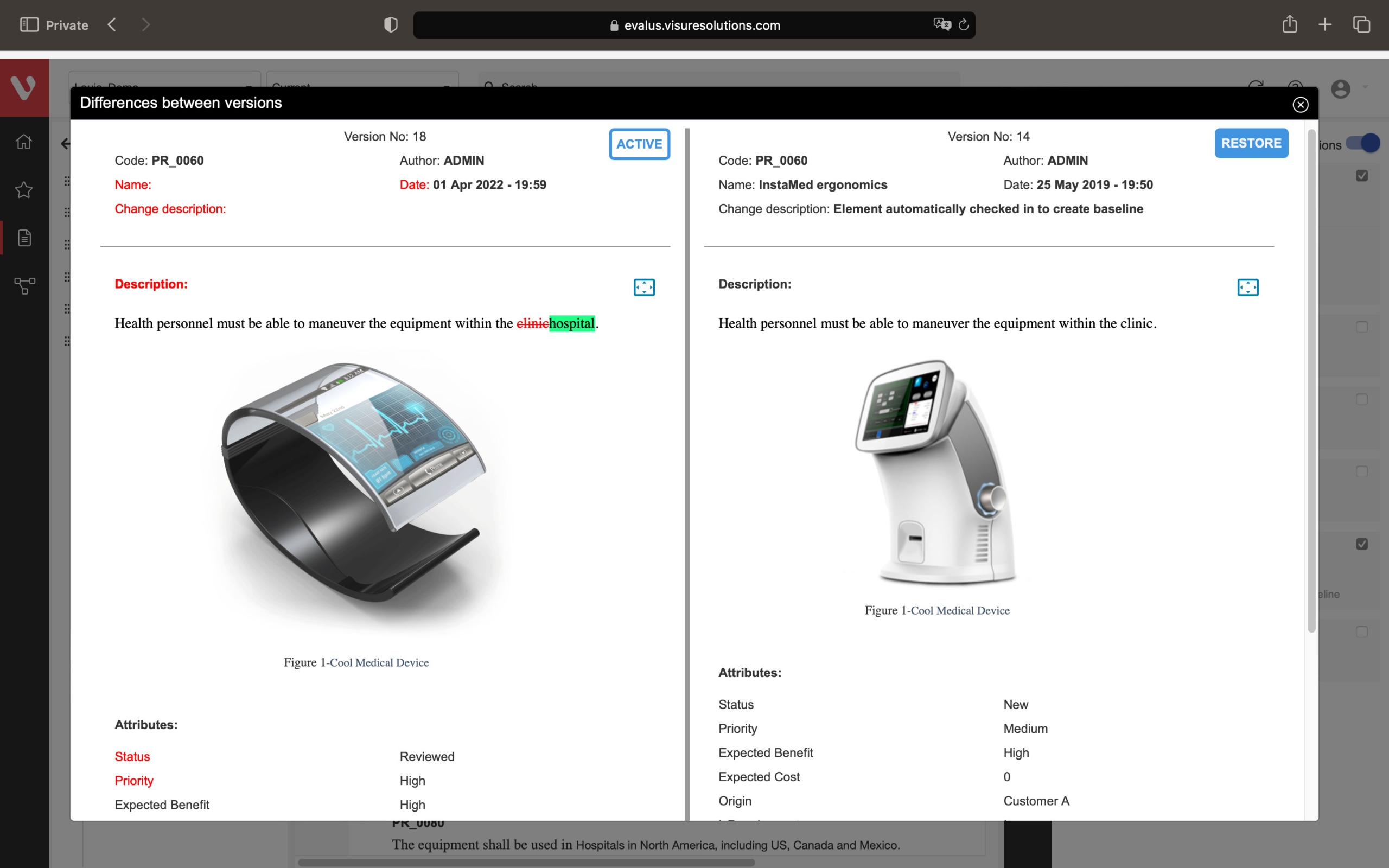
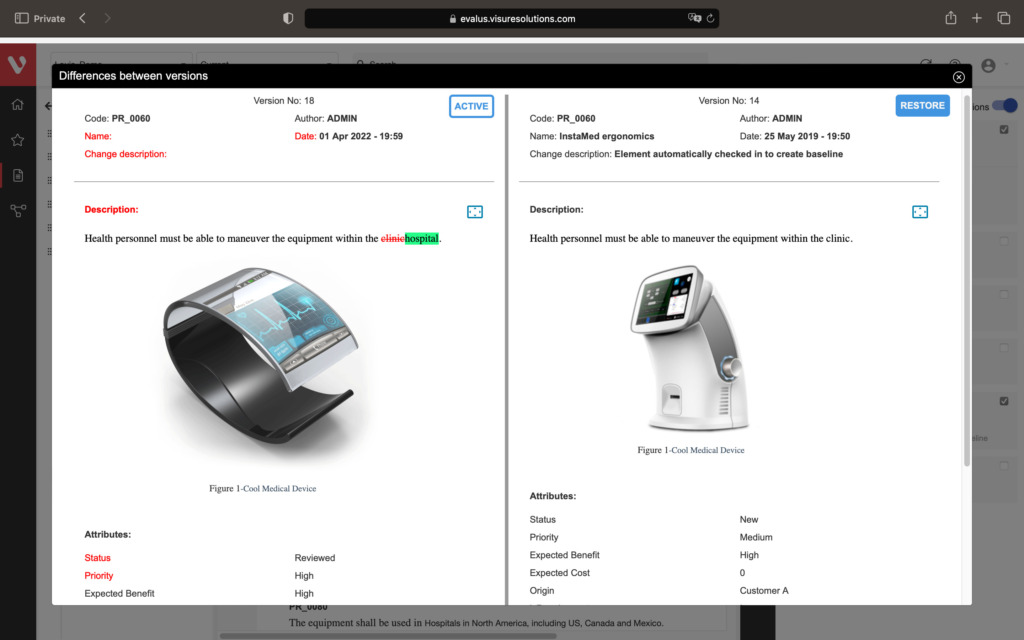
Speed up development and maintain consistency. Easily compare requirement versions, organize and secure your data, and create development stream baselines or requirement catalogs.
Compare changes in requirements, ensure data organization and security, snapshot project states, build reusable requirements catalogs, and develop product variants or new versions.
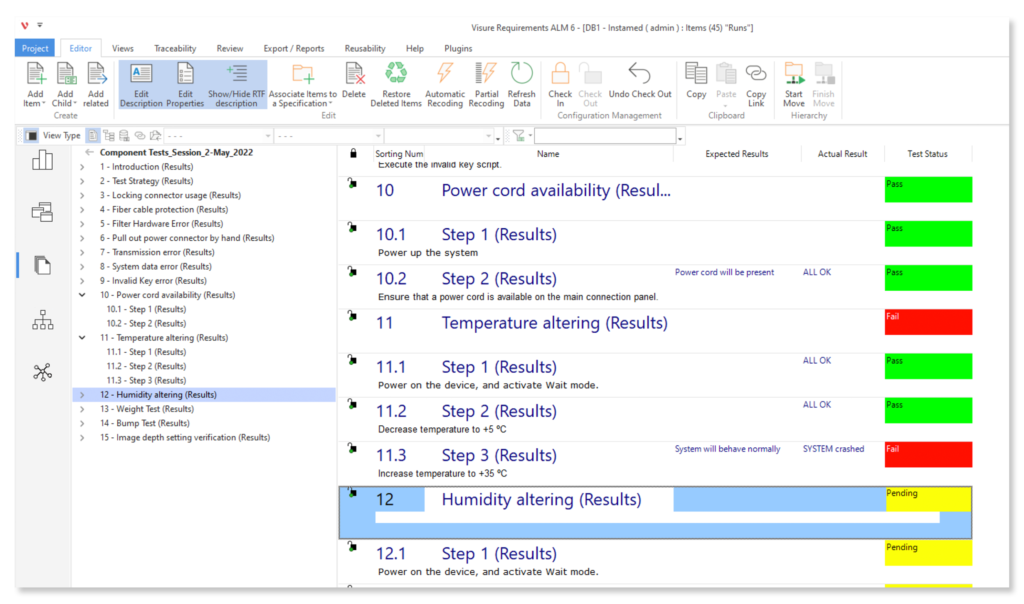
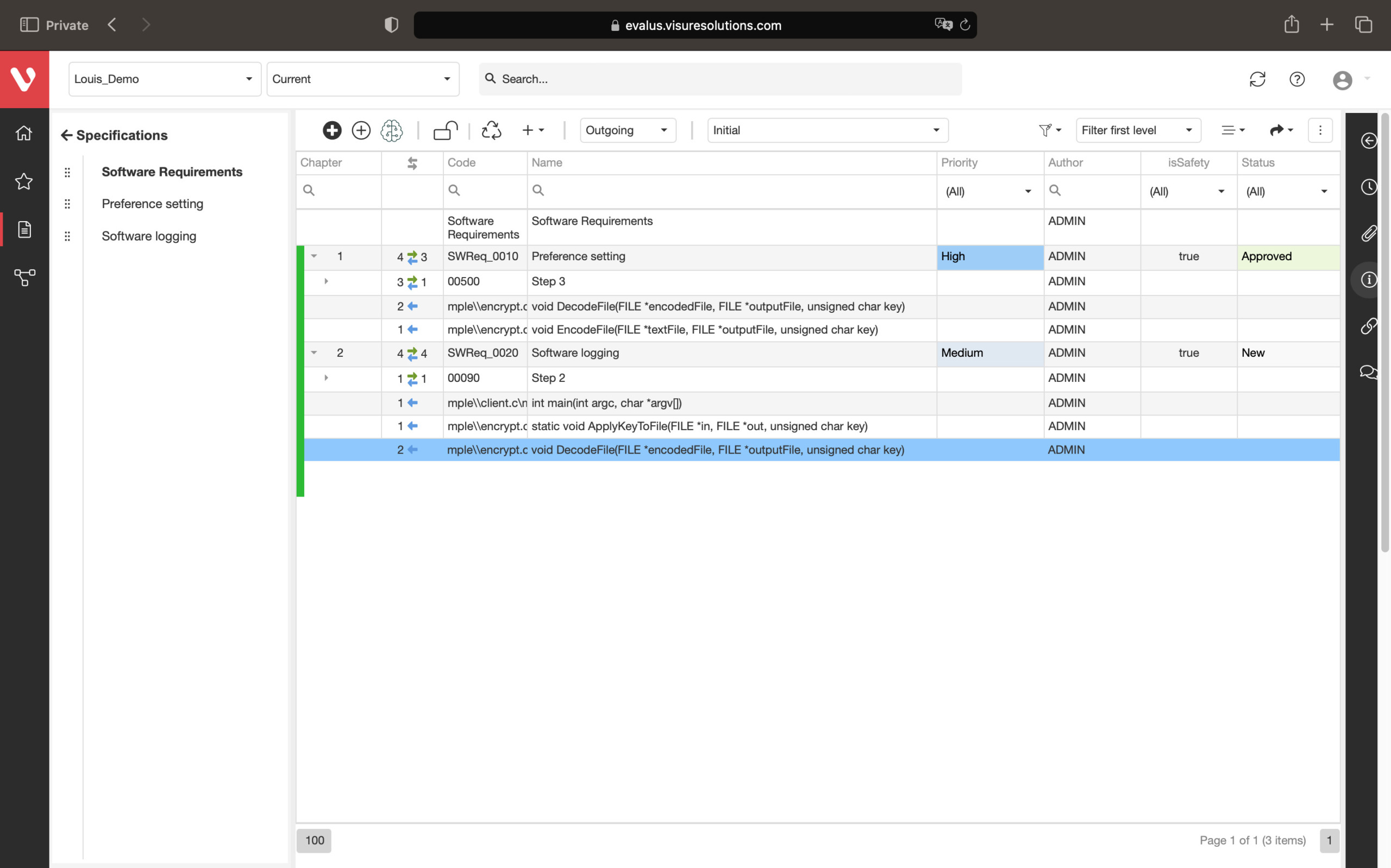 Effortlessly manage real-time testing. Create, organize, and execute plans, cases, and reports. Log defects instantly, prioritize and discuss in real-time. Connect tests, integrate tools, and produce tailored compliance reports for auditors.
Effortlessly manage real-time testing. Create, organize, and execute plans, cases, and reports. Log defects instantly, prioritize and discuss in real-time. Connect tests, integrate tools, and produce tailored compliance reports for auditors.
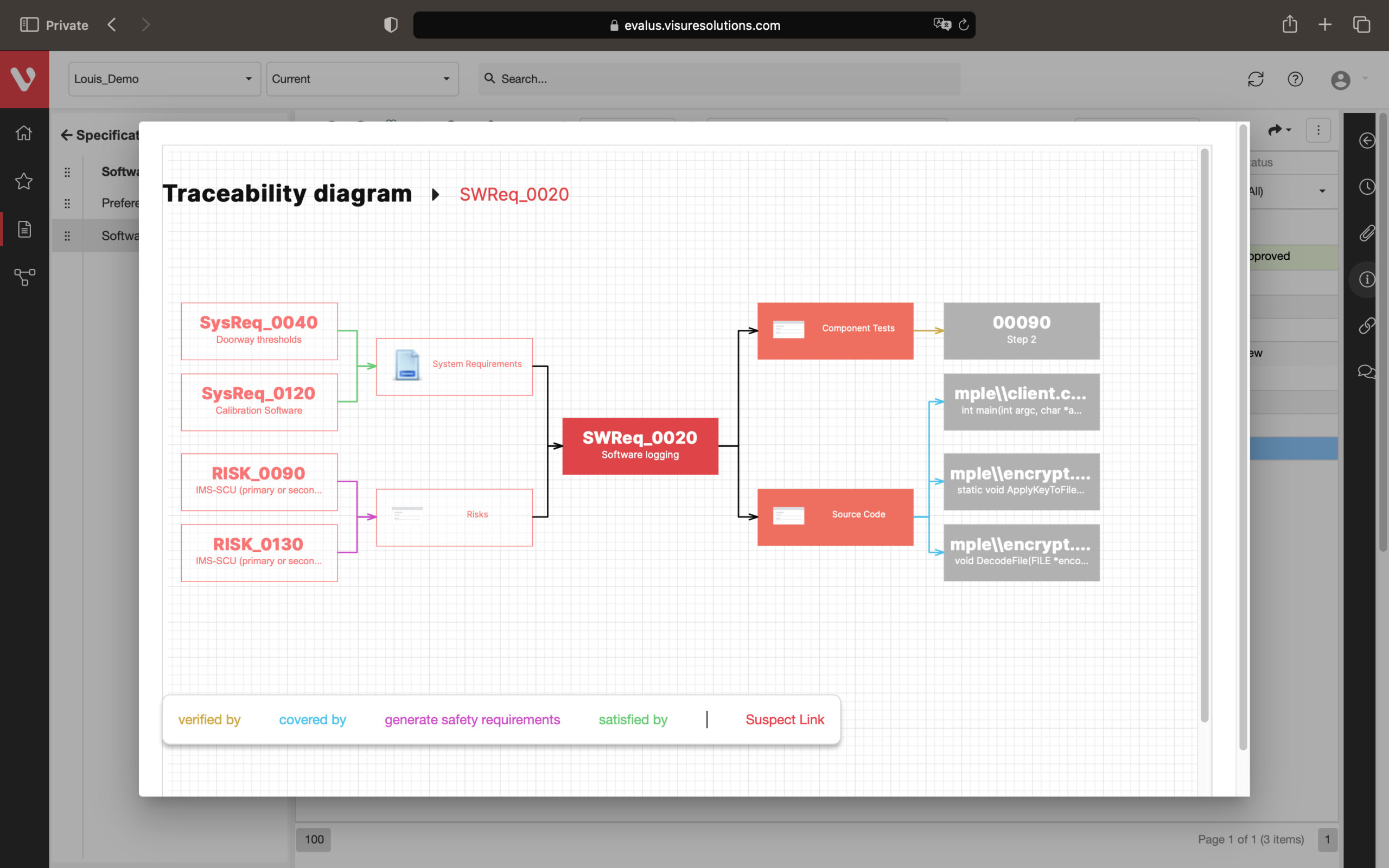
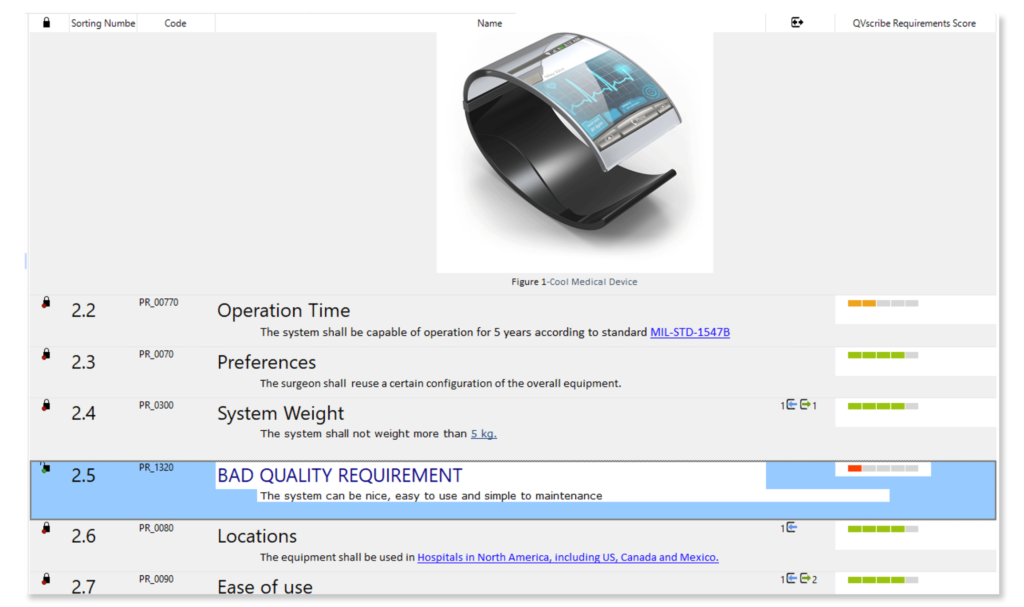
Automatically analyze the quality of requirements while writing them. Avoid ambiguous specifications from poorly written, ambiguous, and inconsistent requirements.
 Ensure secure, cross-functional collaboration across teams, customers, and complex supply chains to remove friction throughout the development process
Ensure secure, cross-functional collaboration across teams, customers, and complex supply chains to remove friction throughout the development process
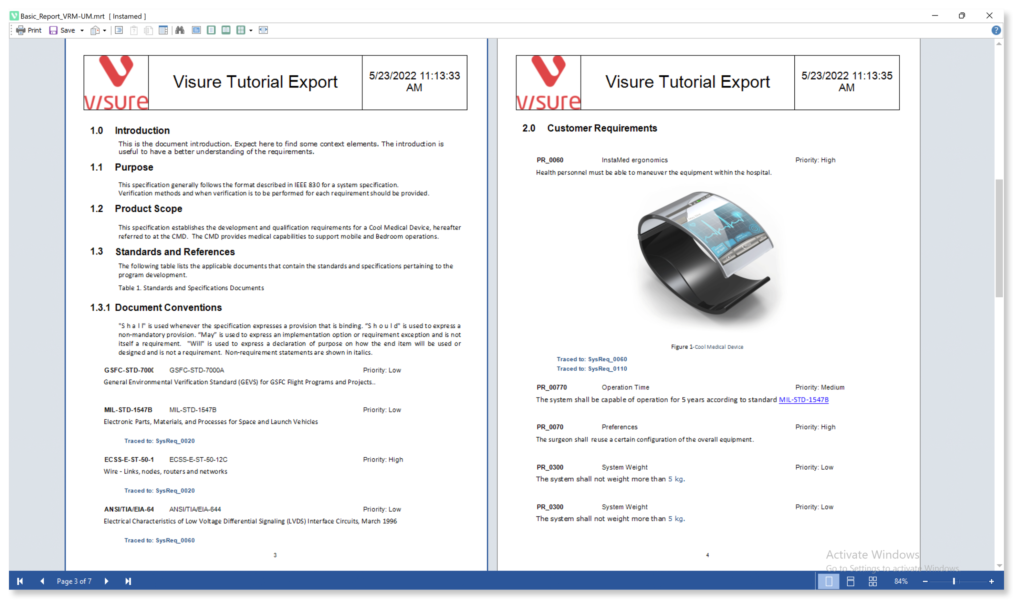
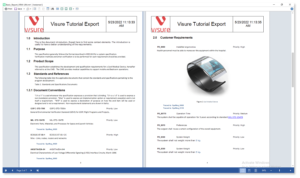 Generate customized documents and reports from your database, helping deliver the necessary regulatory compliance evidence, specifications, test session summaries, or any other required output.
Generate customized documents and reports from your database, helping deliver the necessary regulatory compliance evidence, specifications, test session summaries, or any other required output.
Increase your productivity and ease your Stakeholder review process by using simple import and export data features for ReqIF, and MS Office Word & Excel.

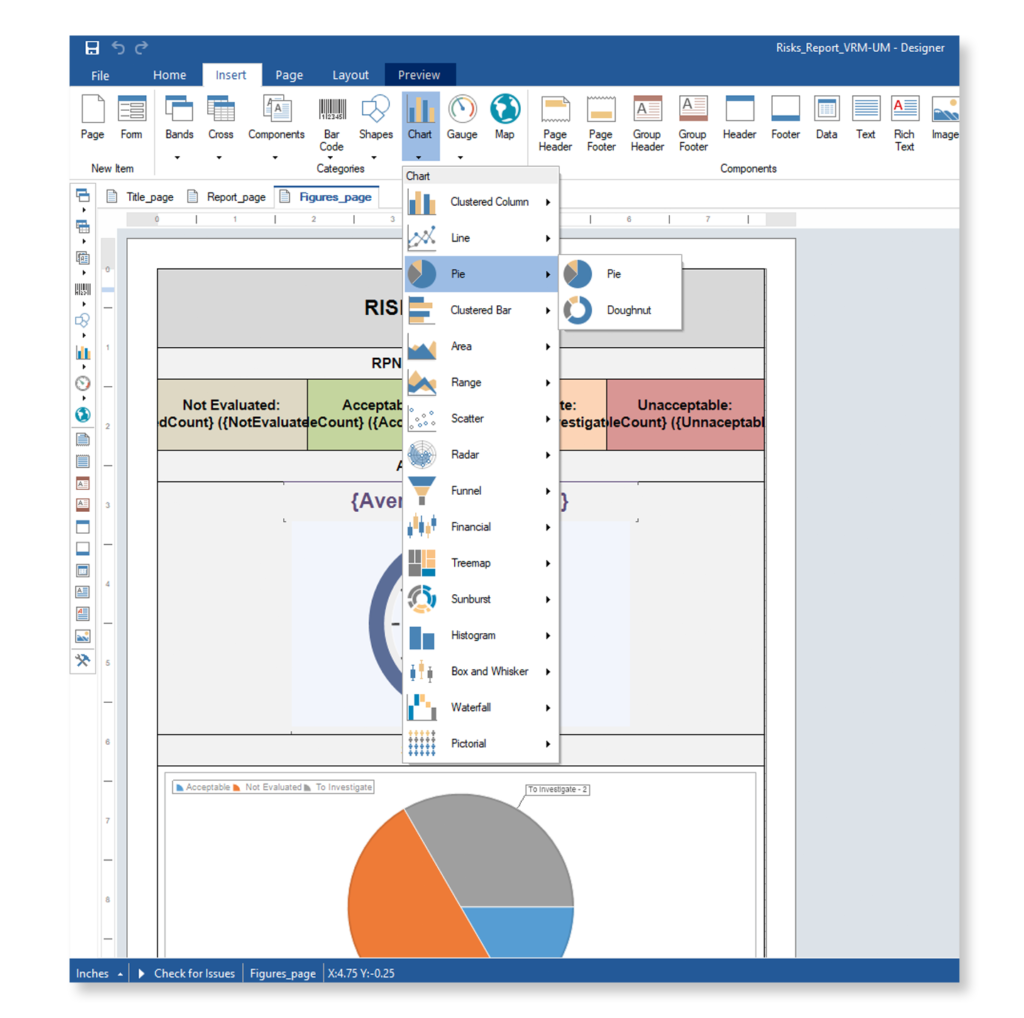
Automate Your Proof of Evidence.
Optimize Your DO-178C & DO-254 Certification Compliance.
With Visure, you can use automated checklists to manage compliance and easily integrate and access our DER partner’s checklists into our tool. This will enable you to design and improve a review process around these checklists.

Why Top Leading Pharmaceuticals Companies Choose Us

Reduce Risk & Manage Standard Compliance
Mitigate Risk and avoid stressful compliance audits across projects by centralizing and tracing in a single source of platform.
Full end-to-end Traceability, including Source Code
Configure your data model and gain full traceability between tests, requirements, risk, defects and all items, including source code to specific requirements.

Simple Import and Export Data
Increase your productivity by using simple import and export data features from ReqIF and MS Office Word and Excel.

Facilitate Real Time Collaboration & Alignment
Visure integrates bi-directionally and automatically with the top industry engineering tools, easing collaboration among teams in real time.

Easy to use UX/UI Requirements ALM Tool
Forget about legacy tools user friendly experience, and implement an easy to use Requirements ALM tool with a low learning curve.

Most Value to Price Product in the Market Guaranteed
We are committed to your team's project success by delivering within budget. That's why Visure's pricing is a fraction from other competitors.

Maintain Security Across Development
With our On-Premise Licensing option, you can easily deploy and maintain security across all your projects within the tool.

Accelerate Project Speed to Market
Increase your team's productivity with reusability of components across projects and automating repetitive tasks through open source code & AI.

Access Premium Support, Trainings and Consultations
Fast track your team's success by getting your team up and running easily, while staying on top with industry best practices.
Visure Requirements ALM Connects with Best-of-Breed Tools
And even more integrations with other leading software — including automated test solutions— to accelerate and facilitate success across the entire product development lifecycle.
What industry professionals say about us



As posted in G2, SoftwareReviews and TrustRadius.
Ensure Pharma Compliance.
Enforce Full Traceability.
Accelerate Your Timelines.
- Most cost-effective
- Access All Features
- 30-Day Trial
On average, our customers experience:
See what’s possible with a Modern Pharma ALM Software Solution
PER PROJECT
TO MARKET
PREPARING FOR AUDITS
FAQs
What is included in Visure's Free trial?
During your free trial, you will have access to Visure’s key tool features, which includes: Requirement Management, Test management, Risk management, Baseline, Reusability, Version control, Review, Dashboard, Traceability Matrix, Impact Analysis Review, and Collaboration.
What is Visure’s Requirements ALM Pricing?
Great question! At Visure, we’ve been ranked as the most affordable modern requirements management tool, providing the most value to price solution in the market. Our pricing varies from company to company due to the different types of licenses and add-ons each one needs. Once we receive a couple of details regarding your needs, we will be able to provide you with an accurate pricing.
What types of licenses are available with Visure Requirements ALM?
We have two main types of licenses: Read & Write License and Read Only License. To Learn more about each type of license, please go over to our Pricing Page.
Do I need a credit card to get started?
No, neither credit cards nor contracts are required for your free trial.
You can test drive Visure, and cancel anytime.
Do I need to sign a contract to get started with my Visure free trial?
No, we don’t require any contracts to get started with Visure’s 30-day free trial.
What Add-ons are available for Visure Requirements ALM Platform?
- To complete the entire Application Development Lifecycle Management, we’ve developed Add-ons for Visure’s core Requirements ALM product. You can learn more about each add-on by visiting each of their pages. Our Add-ons include:
- Automated Checklists
- Report Manager
- Contributor
- Tool Qualification Package
- Quality Analyzer
- All Integrations
What integrations are available with Visure Requirements ALM Platform?
At Visure, we support and have native integrations with: VectorCAST, Jira, Azure DevOps, GitLab, Sparx EA, Word & Excel, and ReqIF. These native integrations are bi-directional, syncing the information automatically between tools. Your team can customize and configure the tool and its integration to fit your workflow methodologies, adapting to processes such a Waterfall, Agile, and Hybrid types of processes
In addition, with ReqIF, engineering teams can exchange data and documents across third party tools. These includes tools such as MATLAB Simulink, Cameo, Rapita Systems, IBM DOORS and Ansys Scade.
If we currently have another Requirements Management & ALM Tool, does Visure support migrations?
By working with 100+ global organizations, we learned that many are stuck with legacy tools and even tools that don’t completely satisfy their needs. For these reasons, at Visure we are committed on giving additional migration support and training when migrating to any of our tools.
Additionally, our ReqIF integration enables teams to easily migrate data between any other tool and Visure.
Where do I contact for Product Support?
If you’re an existing client and require product support, please visit our support page. In addition, if you’re currently evaluating Visure Requirements ALM Platform or any of our Add-on products, we’re committed on giving you any product, technical and customer support.
Does Visure offer tool trainings?
Great question! Yes, we do offer different levels of tool trainings.
