#1 REQUIREMENTS REVIEW SYSTEM SOLUTION
Real-time Access for Every User Level in Your Review Process
Empower every experience level user within your team by customizing their access across the project.
- Most cost-effective
- Access All Features
- 30-Day Trial


1,000+ Highly Regulated Organizations Trust Visure














What is Requirements Review & Why is it important?
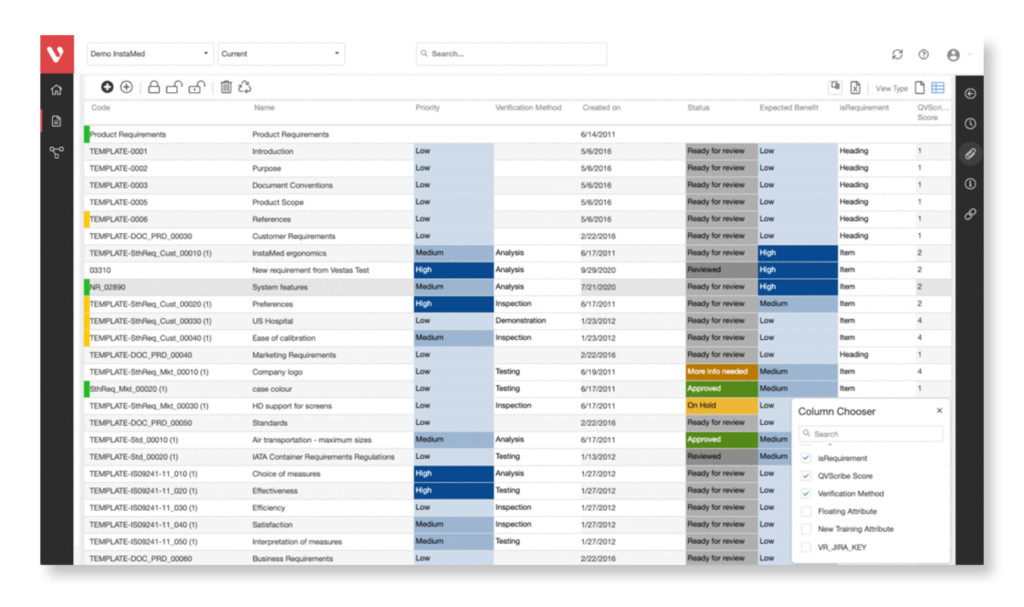
A requirements review process is a systematic approach that involves engagement with key stakeholders and the project team. This review process entails a detailed evaluation of the requirements document line by line.
Requirements authoring is a monotonous and complex process, making it inevitable that errors may occur at some stage. That’s why it’s important to have a requirements review process in place to identify areas that require modifications, corrections, and clarifications.
- Complete, Precise, & Clear Requirements
- Make Better Decisions With Your Team
- Enhanced Change Management
- No Contradictions/Gaps In Requirements
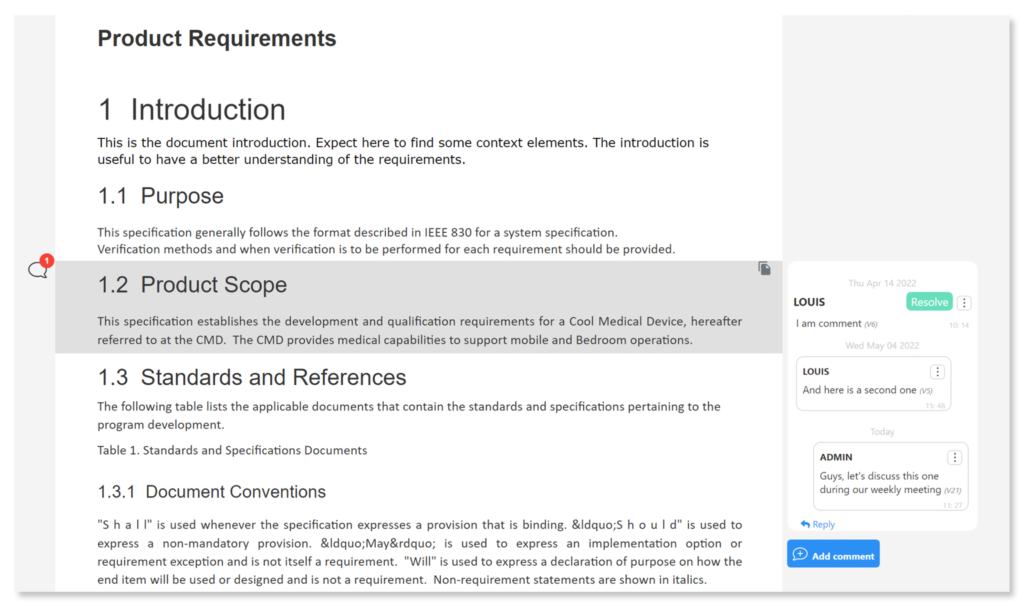
Engage Team Before Implementing Change with Visure's Comments
With Visure’s Commenting feature, you can now involve all stakeholders and team members in every discussion before applying any changes to projects.
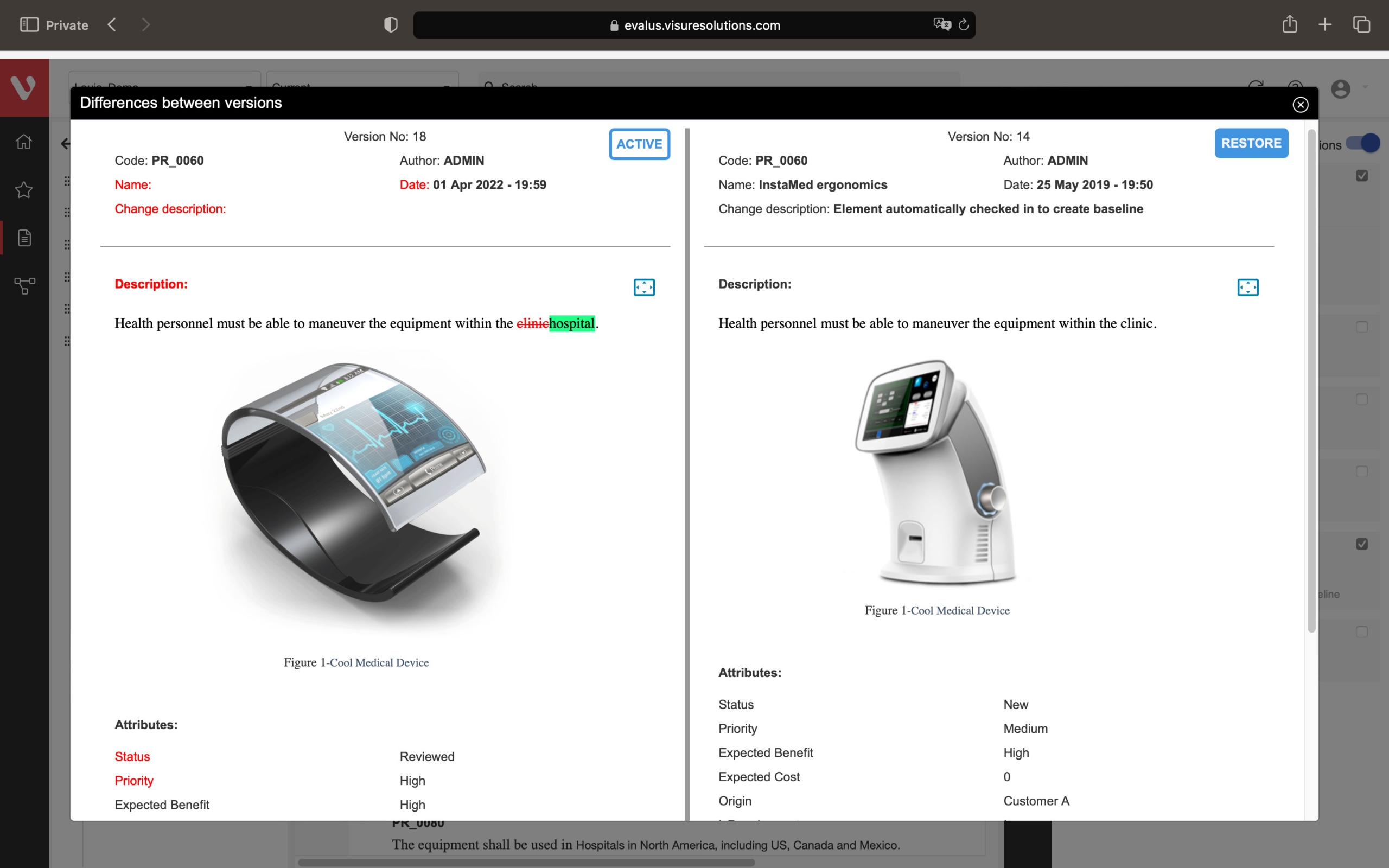
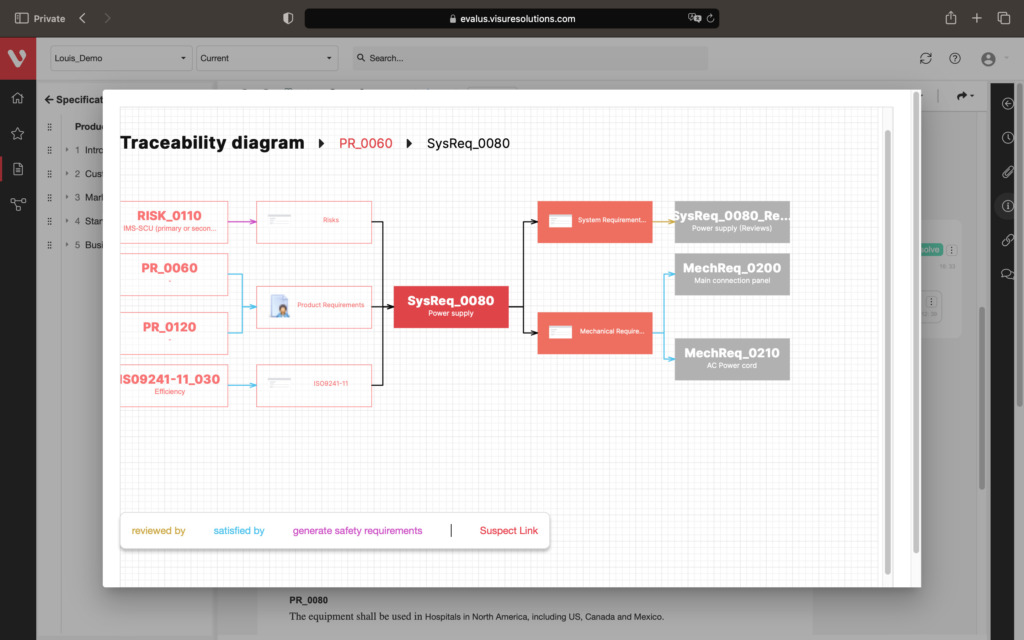
Every time a new version has been created, it triggers the tool to automatically create suspect links which are traced to all of the elements which have been modified.
- Apply Enhanced Change Management
- Suspect Link Traceability With All Elements
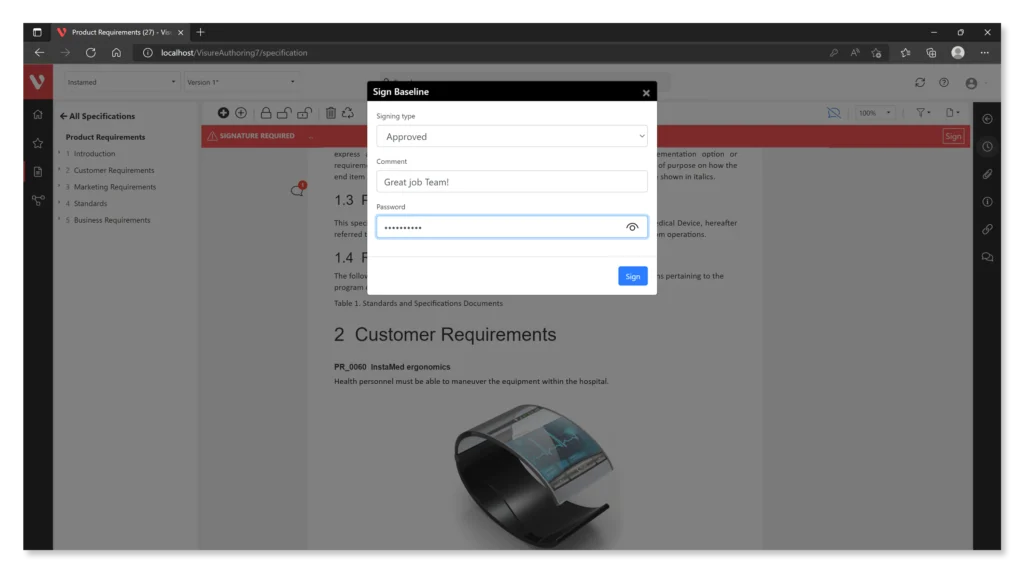
- Create and Adapt Your Workflow
- Automate Baseline Signature Process
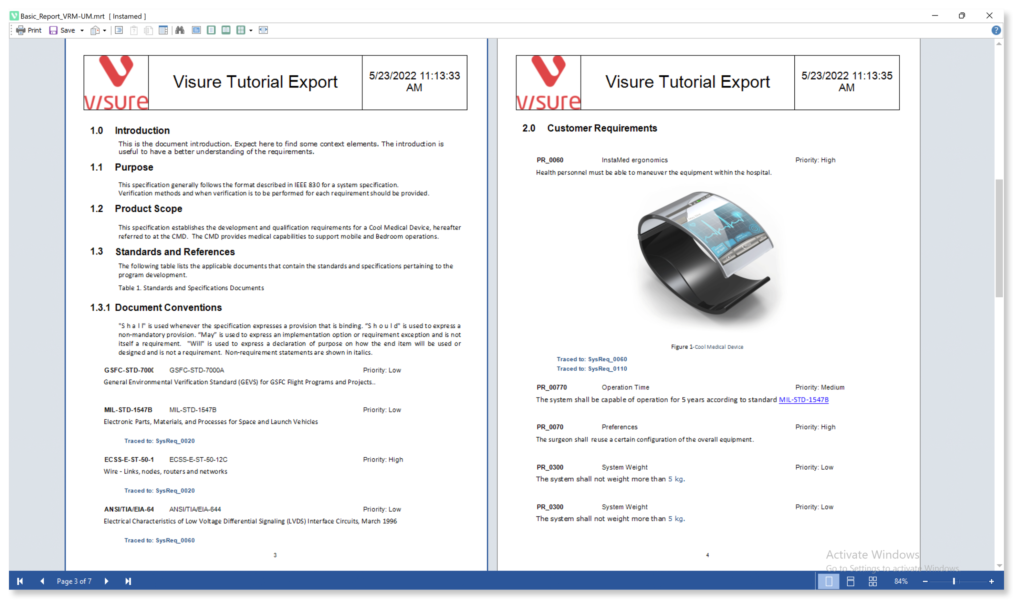
- Export In Any Format (PDF, MS Office, etc)
- Enable Email Alerts & Notifications
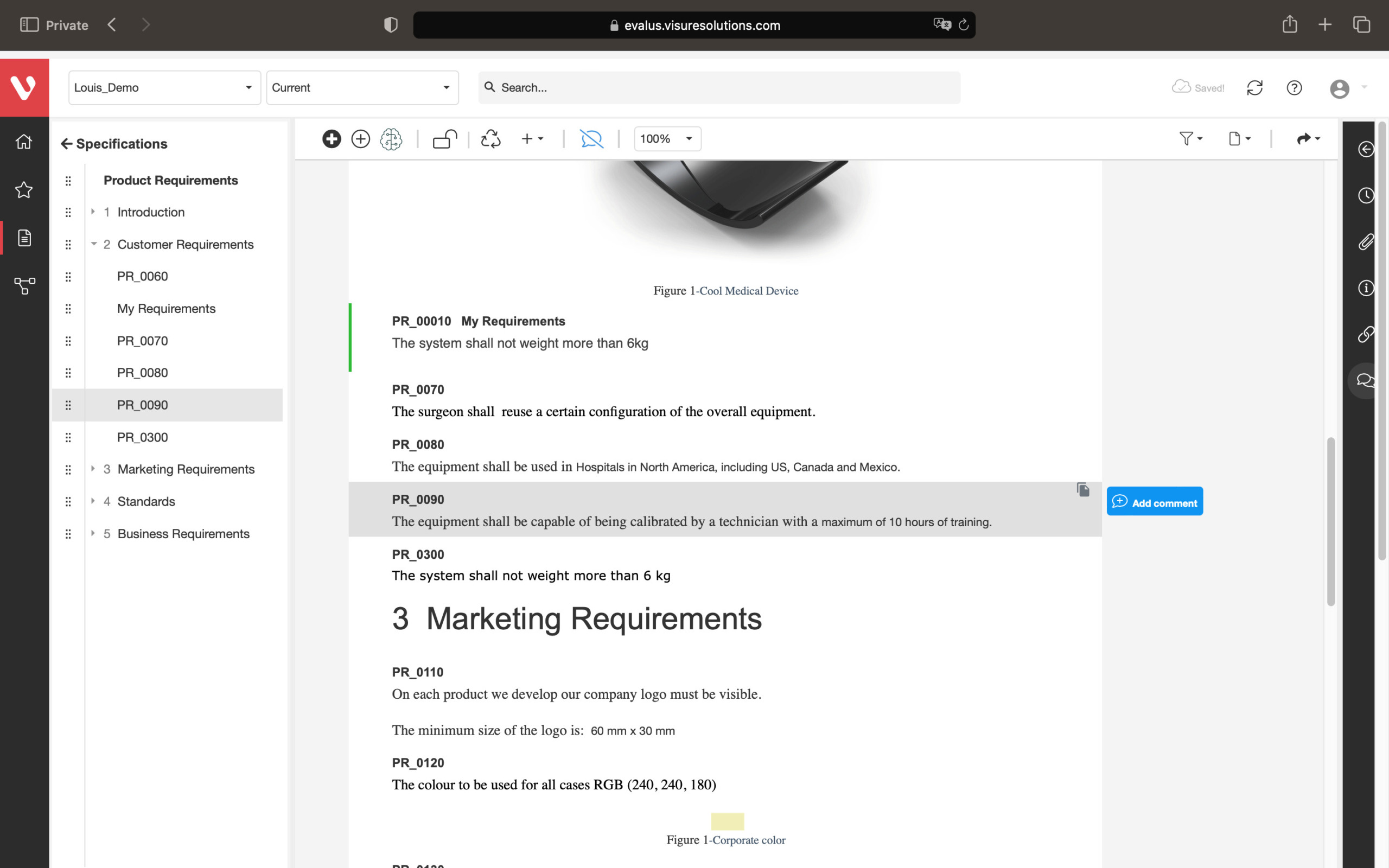
Streamline Your Product Development Process with All-in-One Requirements Review System

Achieve a balance between atomic requirements management and traceability, all while maintaining a familiar document-style approach.

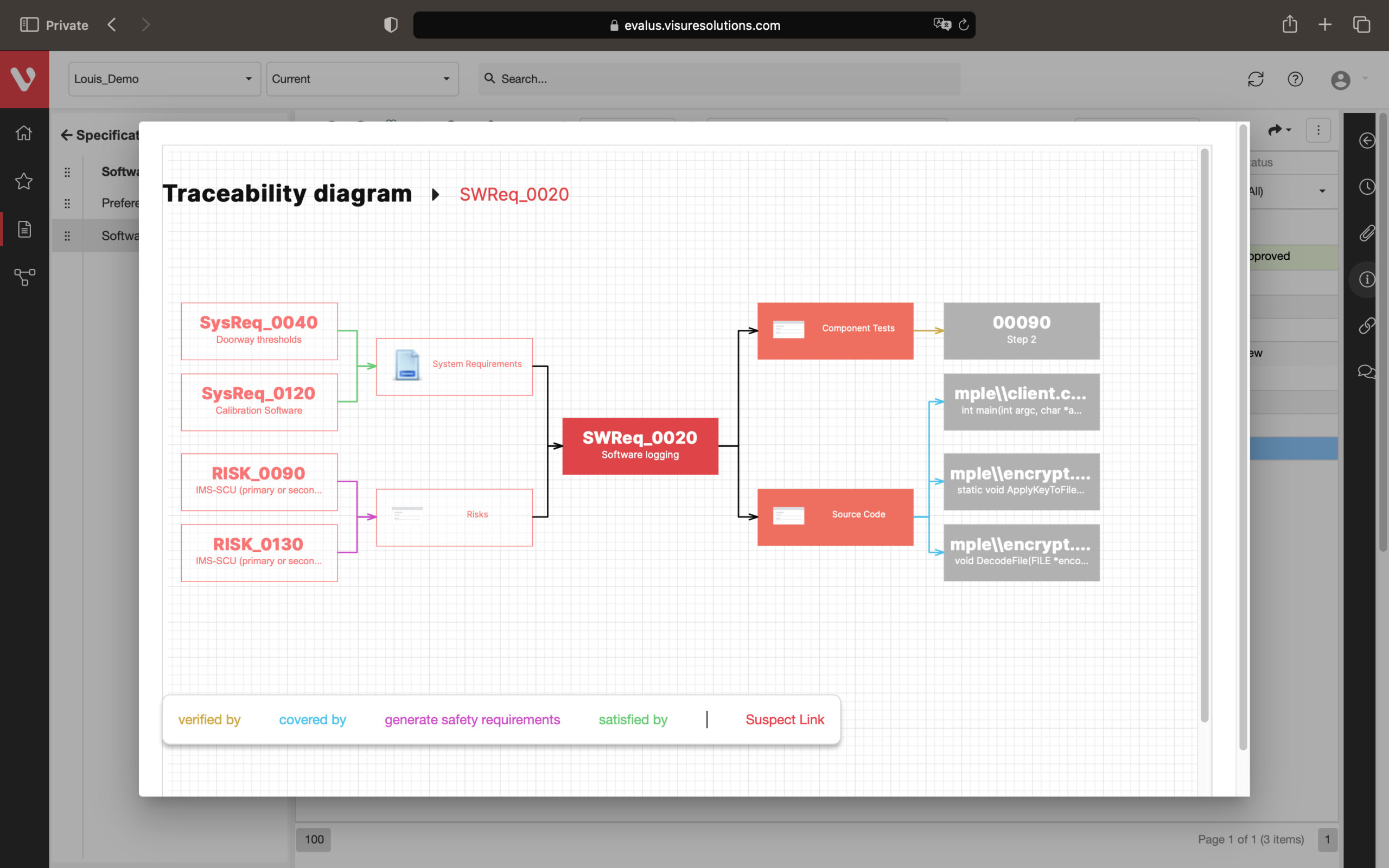
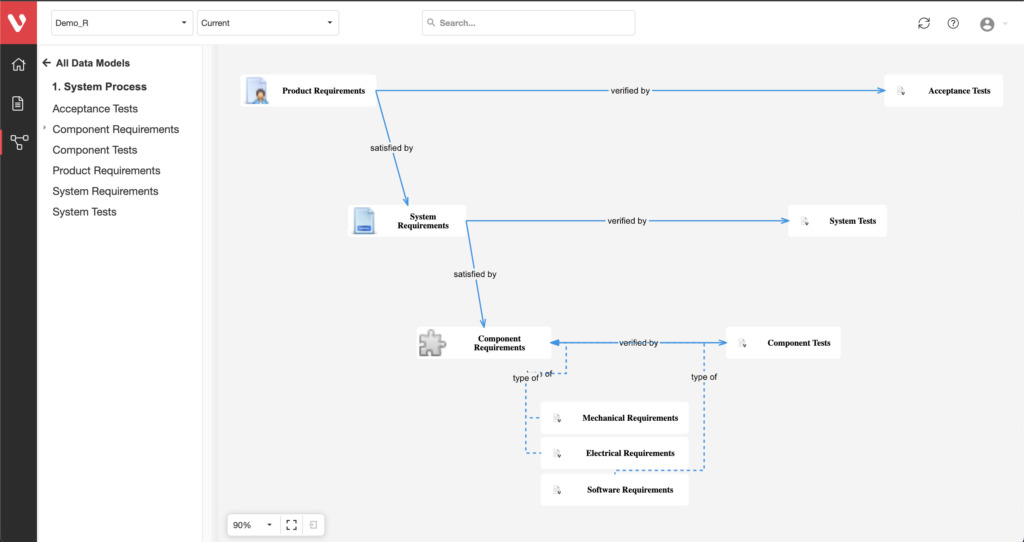
Effortlessly explore upstream and downstream relationships, anticipate change impact, monitor cross-project relationships, detect potential link issues during changes, and visualize relationship rules across projects to understand their organizational impact and reach.
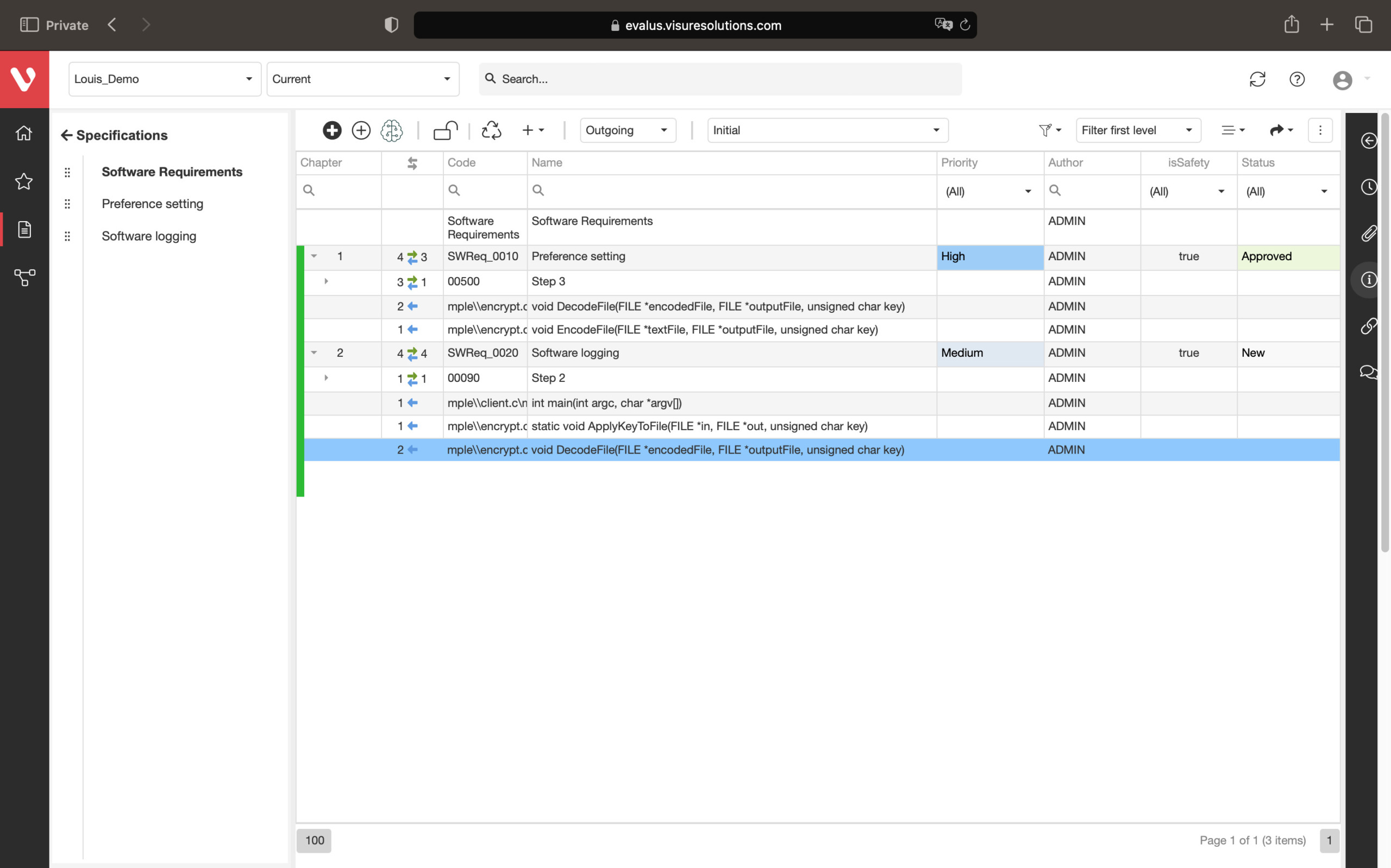
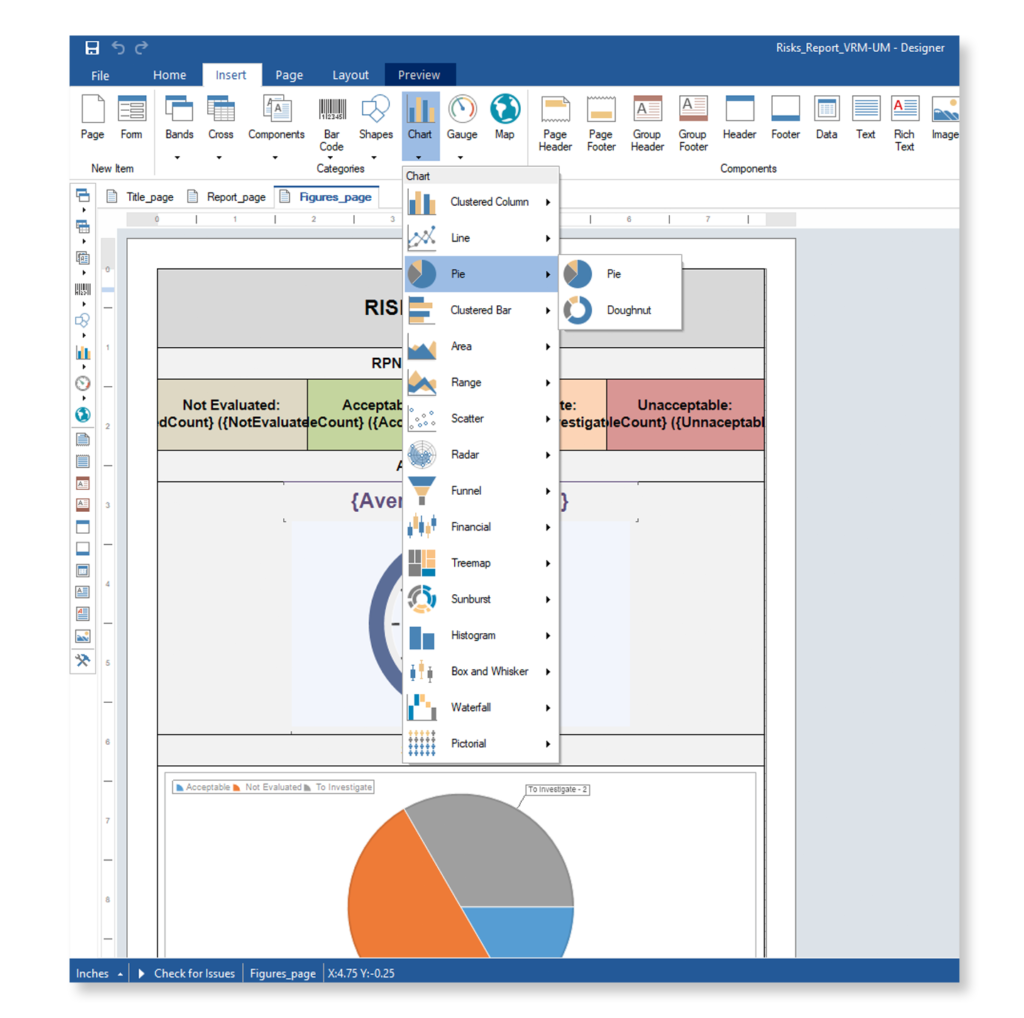
Keep your risk analysis current with real-time data, improve coverage by tracing open risks to requirements, build a comprehensive risk management profile using PHA and FMEA techniques, and guarantee quality and safety in intricate product development.
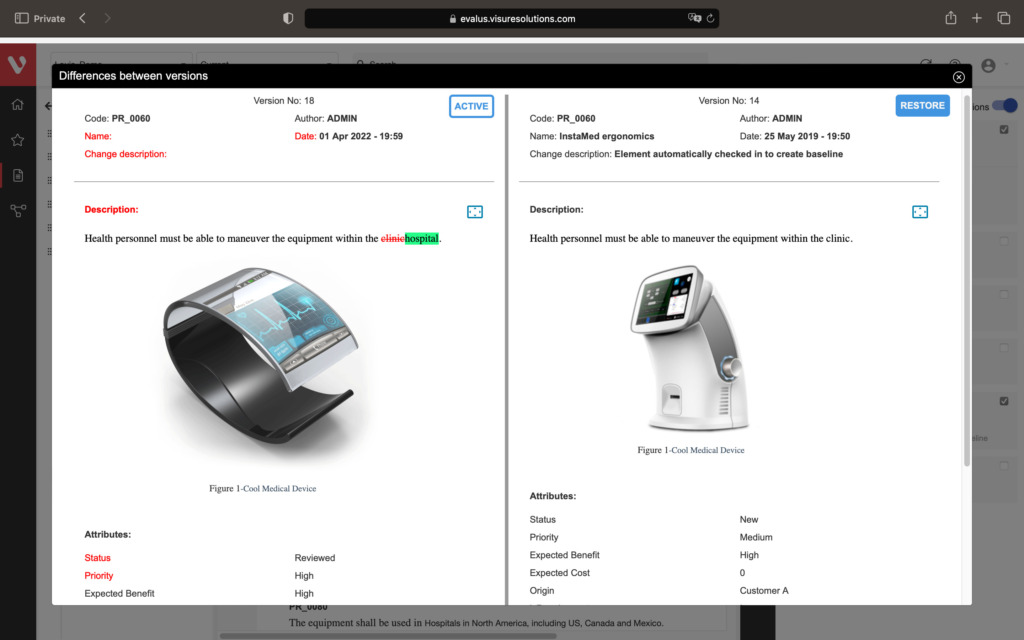
Speed up development and maintain consistency. Easily compare requirement versions, organize and secure your data, and create development stream baselines or requirement catalogs.
Compare changes in requirements, ensure data organization and security, snapshot project states, build reusable requirements catalogs, and develop product variants or new versions.
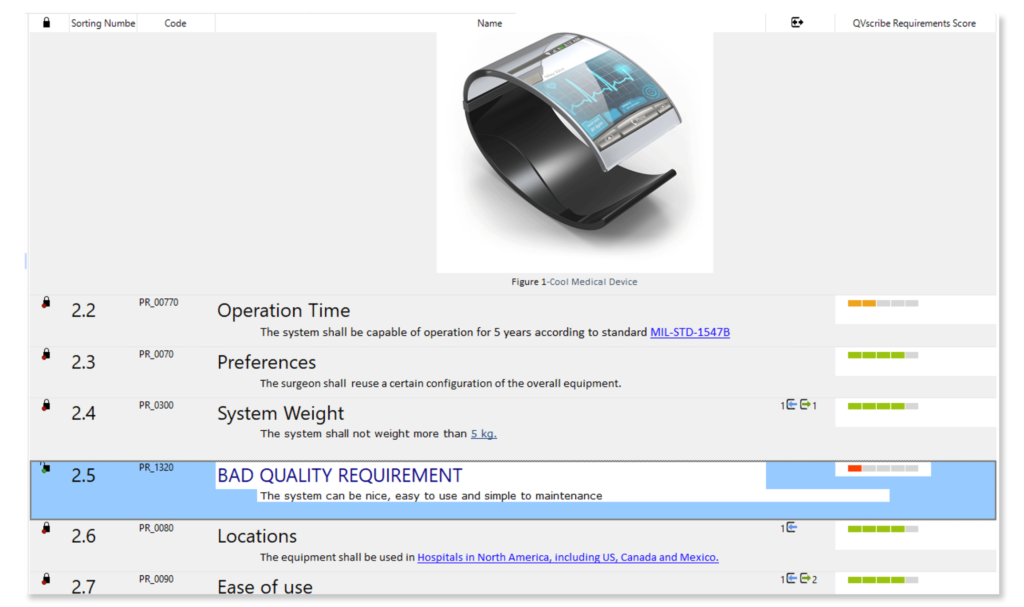
Automatically analyze the quality of requirements while writing them. Avoid ambiguous specifications from poorly written, ambiguous, and inconsistent requirements.

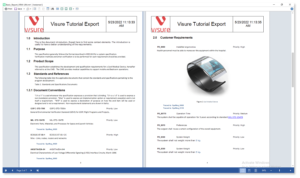
Increase your productivity and ease your Stakeholder review process by using simple import and export data features for ReqIF, IBM DOORS and MS Office Word & Excel.
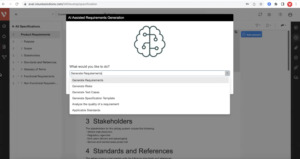
Leverage generative AI to analyze and optimize requirements writing and industry best practices, generate test cases and improve risk mitigation

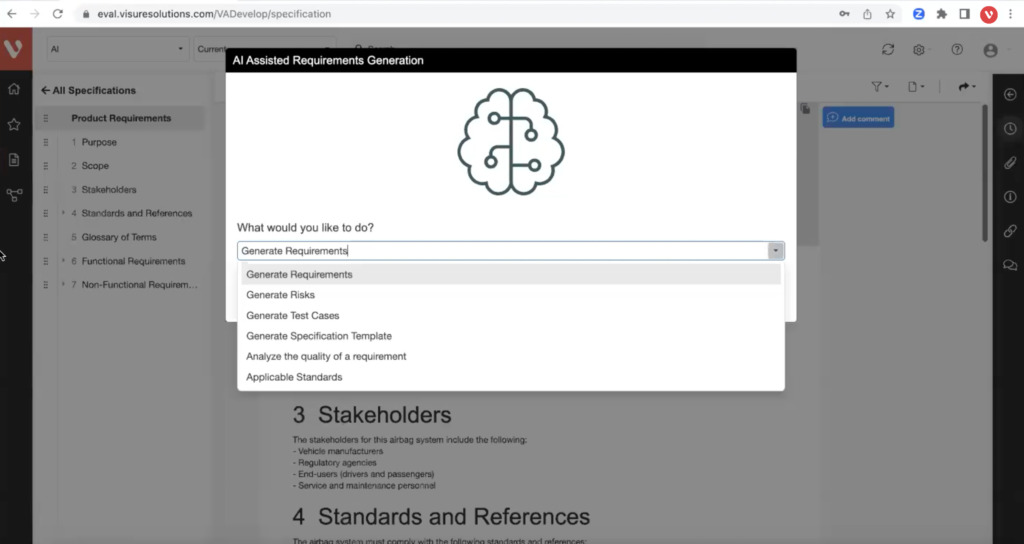
Why Top Leading Safety Critical Companies Manage & Review Requirements with Visure

Reduce Risk & Manage Standard Compliance
Mitigate Risk and avoid stressful compliance audits across projects by centralizing and tracing in a single source of platform.
Full end-to-end Traceability, including Source Code
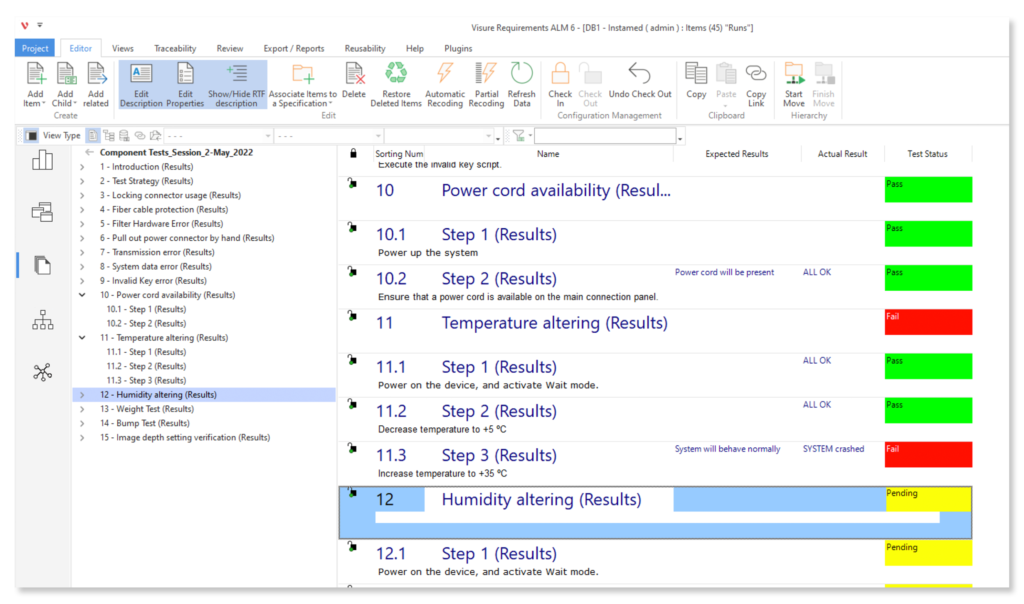
Configure your data model and gain full traceability between tests, requirements, risk, defects and all items, including source code to specific requirements.

Simple Import and Export Data
Increase your productivity by using simple import and export data features from ReqIF and MS Office Word and Excel.

Facilitate Real Time Collaboration & Alignment
Visure integrates bi-directionally and automatically with the top industry engineering tools, easing collaboration among teams in real time.

Easy to use UX/UI Requirements ALM Tool
Forget about legacy tools user friendly experience, and implement an easy to use Requirements ALM tool with a low learning curve.

Most Value to Price Product in the Market Guaranteed
We are committed to your team's project success by delivering within budget. That's why Visure's pricing is a fraction from other competitors.

Maintain Security Across Development
With our On-Premise Licensing option, you can easily deploy and maintain security across all your projects within the tool.

Accelerate Project Speed to Market
Increase your team's productivity with reusability of components across projects and automating repetitive tasks through open source code & AI.

Access Premium Support, Trainings and Consultations
Fast track your team's success by getting your team up and running easily, while staying on top with industry best practices.
Visure Requirements Management Solution Connects with Best-of-Breed Tools
And even more integrations with other leading software — including automated test solutions— to accelerate and facilitate success across the entire product development lifecycle.
What industry professionals say about us





As posted in G2, SoftwareReviews and TrustRadius.
Ensure Regulatory Compliance.
Boost Collaboration & Efficiency.
Accelerate Your Timelines.
- Most cost-effective
- Access All Features
- 30-Day Trial
On average, our customers experience:
See what’s possible with a Modern Requirements Review Software Solution
PER PROJECT
TO MARKET
PREPARING FOR AUDITS
The Ultimate Guide to Requirements Management & Traceability Best Practices
Hardware and software complexity is rapidly increasing across all highly regulated industries. Learn the best Requirements Management and Traceability practices that leading safety-critical companies use to mitigate risk and to manage product, systems, and software development.